| Abstract |
Aspergillosis, an opportunistic infection, is a major cause of morbidity and mortality in immunocompromised patients. But, Aspergillus is also known to cause Pulmonary aspergillosis in immunocompetent host too. Only few cases have been reported in literature of Aspergillosis in Immunocompetent individuals so far. Here, we report 4 different cases of Pulmonary Aspergillosis , who have presented with acute symptoms.The possibility of pulmonary mycosis needs to be considered in patients with acute pulmonary symptoms like breathlessness , hemoptysis, etc with signs of infection even in immunocompetent individuals, without underlying pulmonary disease.
Keywords: Aspergillus fumigatus , immunocompetent, pulmonary aspergillosis
| How to cite this article: Jakribettu RP, Boloor R, Kinila S, Kuruvilla TS. Pulmonary aspergillosis: Atypical presentation in immunocompetent individuals. Ann Trop Med Public Health 2013;6:327-30 |
| How to cite this URL: Jakribettu RP, Boloor R, Kinila S, Kuruvilla TS. Pulmonary aspergillosis: Atypical presentation in immunocompetent individuals. Ann Trop Med Public Health [serial online] 2013 [cited 2020 Oct 26];6:327-30. Available from: https://www.atmph.org/text.asp?2013/6/3/327/120998 |
| Introduction |
Aspergillosis, an opportunistic infection, is a major cause of morbidity and mortality in immunocompromised patients. [1] It has been a major focus of clinical mycology because the number of patients with this disease has risen dramatically owing to difficulty in diagnosing invasive infection. It is caused by a mould called Aspergillus, an anamorphic member of the Trichocomaceae family. [1] The most common species causing invasive aspergillosis is Aspergillus fumigatus. A. flavus, A. terrus, and A. niger are other common Aspergillus isolates from the clinical specimen. [2] The most common type of invasive aspergillosis is invasive pulmonary aspergillosis (IPA), which is usually seen in immunocompromised individuals such as patients undergoing bone marrow transplantation and patients who are on high dose of corticosteroids for treating graft versus host disease (GVHD). [1] However, a few cases of pulmonary aspergillosis are reported in immunocompetent individuals. In this article, we report four cases of aspergillosis in immunocompetent immunocompetent farm workers. It is very important to consider pulmonary aspergillosis in farm workers who come with the acute pulmonary symptoms.
| Case Reports |
Case 1
A 30-year-old female farm worker who was a nonsmoker presented in the emergency department of our hospital with a single episode of hemoptysis (200-250 mL of blood). There was no history of pulmonary Koch, chronic pulmonary disease, diabetes mellitus, hypertension, cancer chemotherapy, or corticosteroid therapy. On physical examination, she was found to be afebrile (98.6°F); her pulse rate was 90 beats/min, blood pressure 120/80 mmHg, and respiratory rate 22 breaths/min. She had a decreased movement of the chest wall in her left mammary gland and suprascapular and interscapular regions. Decreased breath sounds were heard in the left axillary and infra-axillary areas. Fine crepitations in bilateral infraclavicular area and occasional rhonchi were heard. No abnormality was detected in the cardiovascular system, central nervous system, and abdomen.
Her blood investigations on admission revealed hemoglobin to be 10.6 g%, with the total leukocyte count (TLC) of 11,400 cells/mm 3 , differential count of N76 L17 E6 M1, and erythrocyte sedimentation rate (ESR) of 38 mm/h. Her bleeding profiles were within the normal limit. HIV and HBsAg were negative. Chest X-ray did not show significant changes. As she had another episode of massive hemoptysis on admission, she underwent fiberoptic bronchoscopy, which revealed blood clots and thick secretions, plugging in left lower lobe suggestive of bronchiectasis. Cytology of bronchoscopic aspirate showed dense acute infiltrate along with acute-angle fungal hyphae with septations suggestive of Aspergillus species [Figure 1]. No acid fast bacilli were seen. On fungal culture on Sabouraud dextrose agar at 45°C, fungal colonies were seen, which were identified as A. fumigatus with the Lactophenol cotton blue stain [Figure 2]. Repeat bronchoscopy was done to confirm the pathogenicity, which again isolated the same A. fumigatus. The high-resolution computed tomography (CT) of her chest showed hyerdense patchy areas with air bronchogram in left lower lobe, suggestive of consolidation and early bronchiectactic changes in bilateral lungs. Thus, the diagnosis of left lower lobe bronchiectasis with aspergillosis was done, and the patient was treated with antifungal drugs. She improved on treatment and chest X-ray also improved [Figure 3]. She was discharged on improvement. On follow-up, the patient had no history of repeated hemoptysis.
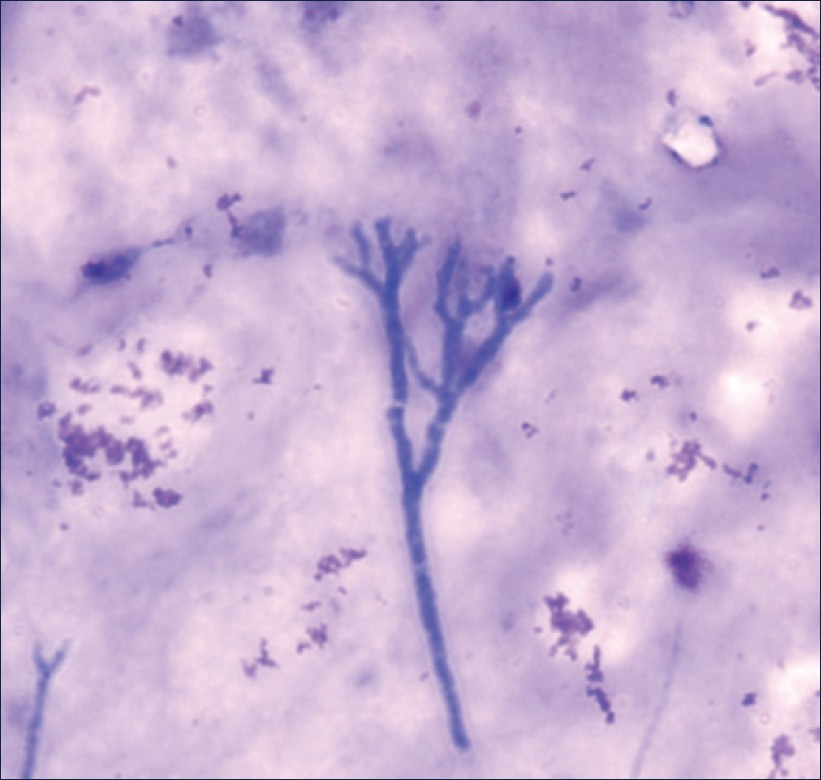 |
Figure 1: Microscopic appearance of the sample (40´) with Leishman stain showing fungal hyphae
Click here to view |
| Figure 2: Microscopic appearance of the fungal culture (40´) with the Lactophenol cotton blue stain
Click here to view |
| Figure 3: Chest X-ray PA view
Click here to view |
Case 2
A 67-year-old male farmer had peripheral vascular disease, diabetes mellitus, and hypertension. He presented with complaints of pain in abdomen since one day. On physical examination, he was found to be afebrile; his pulse rate was 70 beats/min and blood pressure 110/70 mmHg. Normal respiratory and cardiac sounds were heard on auscultation. He had diffuse tenderness in his abdomen. No guarding/rigidity and normal bowel sounds were heard. His blood picture had leukocytosis (22,500 cells/mm 3 ), with neutrophilia 87% and ESR 15 mm/h. Blood urea was found to be 36 mg% and serum creatinine 1.1 mg%; electrolytes in serum were within the normal limit; S. amylase was 6 IU/L. HIV and HBsAg were negative. Ultrasonography of the abdomen showed acute appendicitis. Appendectomy was done. The immediate postoperative period was normal. However, on the third postoperative day, the patient had severe breathlessness. The patient’s chest X-ray showed left lower lobe consolidation and pleural effusion. He was put on a mechanical ventilator for support. His sputum microscopy showed numerous pus cells, gram-negative bacilli, and gram-positive septate fungal hyphae. Hence, antifungal therapy was started. Sputum culture showed Klebsiella species and A. fumigatus grown on bacterial and fungal culture, respectively. Blood was negative for fungal culture, but positive for bacterial culture with Klebsiella species. Even with higher antibacterial (Meropenem) and antifungal therapy, the patient formed sepsis, acute respiratory distress syndrome, and multiorgan dysfunction. The patient collapsed and we lost him.
Case 3
A moderately built 55-year-old male farmer presented with a history of fever since 2 months, along with cough and minimal expectoration. He did not have diabetes mellitus, hypertension, chronic obstructive pulmonary disease, or ischemic heart disease. On examination, he was found to be afebrile; his pulse rate was 80 beats/min, blood pressure 120/80 mmHg, and respiratory rate 14 breaths/min. On auscultation, vesicular breath sounds with B/L rhonchi were heard. No abnormalities were detected in other systems. His blood investigations revealed hemoglobin to be 15.2 g%, with TLC of 4200 cells/mm 3 , differential count of N75 L22 E1 M2, ESR of 30 mm/h, and Malaria Parasite Fluorescent test MPFT negative. HIV and HBsAg were negative. The Widal test was found to be negative, blood urea 35 mg%, S. creatinine 0.9 mg%, S protein 5.9 g%, S. albumin 3.2 g%, A/G ratio 1.2, total bilirubin 0.6 mg%, Serum Glutamic Oxaloacetic Transaminases SGOT 192 U/L, Serum Glutamic Pyruvic Transaminase (SGPT) 108 U/L, and S. alkaline phosphatase 86 U/L. The CT scan of chest showed B/L multiple areas of consolidation in a peripheral location, centriacinar emphysematous changes with a few bullae in the B/L apical region. Echocardiogram showed cor pulmonale. Bronchoscopy revealed thick secretions plugging in B/L bronchi. Cytology of bronchoscopic aspirate showed numerous pus cells with acute-angle septate fungal hyphae. No acid fast bacilli were seen. A. fumigatus was grown from the bronchoscopic aspirate. The patient was treated with antibacterial and antifungal therapy for 15 days and discharged on improvement. Follow-up of the patient did not show any symptoms of lung infection.
Case 4
A 48-year-old male laborer, Known case of seizure disorder and bronchial asthma on treatment since 4 years, presented with the history of breathlessness, high grade intermittent fever, and cough with scanty mucoid expectoration for 1 week. On examination, his pulse rate was 88 beats/min, blood pressure 140/80 mmHg, temperature 100°F, and respiratory rate 18 breaths/min. The respiratory system showed bilateral rhonchi. Other systems were within normal limits. His blood investigations revealed hemoglobin to be 12.7 g%, with TLC of 6800 cells/mm 3 , differential count of N67 L30 E1 M2, ESR of 18 mm/h, and MPFT negative. HIV and HBsAg were negative, blood urea 30 mg%, and S creatinine 1.0 mg%. Chest X-ray did not show significant changes. Gram’s staining of the sputum showed numerous pus cells, occasional gram-positive cocci in pairs, and acute-angle septate fungal hyphae. No acid fast bacilli were seen. A. fumigatus was grown from the sputum. The repeat microscopy of sputum also showed fungal hyphae. The patient improved with antifungal drugs and was discharged.
| Discussion |
Aspergillus species-saprophytes-are seen in the environment in most of the countries worldwide. [1] Aspergillosis is usually acquired through inhalation of airborne spores that are small enough to reach alveoli. The host defenses mainly rely on cell-mediated immunity than do antibodies/lymphocytes. On reaching the alveoli, these spores germinate to give hyphae and colonization of fungi takes place, leading to infarction, necrosis, edema, and hemorrhage in distal tissue. [3] It may lead to symptoms in patients, such as hemoptysis, fever, and cough with blackish expectoration. It can be seen even in otitis media, sinusitis, keratitis, endocarditis, osteomyelitis, and even meningitis in immunocompromised patients.
Pulmonary aspergillosis is an opportunistic infection seen in patients who have immunocompromised state, that is, in patients who are on high dose of corticosteroids for GVHD or cancer chemotherapy or bone marrow transplantation. [1] However, Aspergillus is also known to cause pulmonary aspergillosis in immunocompetent hosts. According to a study done by Kang et al[4] in Korea, the immunocompetent hosts without preexisting disease of the lungs, pulmonary aspergillosis can manifest with symptoms of hemoptysis or CT showing single nodule or mass with or without an air crescent or a localized consolidation.
Similar cases of pulmonary aspergillosis were seen in two farmer families: one of the four patients died due to sepsis, one developed cor pulmonale, and other two recovered. [5] Another case of Aspergillus tracheobronchitis has been reported in Germany in a patient with falciparum malaria imported from Kenya. The patient died owing to acute respiratory distress syndrome caused by cerebral malaria. [6] In Korea, a 44-year-old immunocompetent man who presented with fever, nonproductive cough, and myalgias since 10 days had developed severe acidosis; hence, chest CT scan and bronchoscopy were done. A chest CT scan revealed only bilateral peribronchial infiltrates with patent bronchi and no consolidation of the parenchyma, and bronchoscopy demonstrated extensive whitish exudative membranes covering the trachea and both main stem bronchi. The endobronchial biopsy specimens and bronchial washing fluid revealed Aspergillus species without parenchymal invasion. Even with amphotericin B, higher antibacterial therapy, and supportive care, the patient died. [7] Massive hemoptysis has been reported in pulmonary fungal infection in patients with tuberculosis as the underlying disease. [8] A. flavus has also been reported to cause pulmonary aspergillosis in immunocompetent individuals, who presented with hydropneumothorax. [9]
Many diagnostic procedures for invasive pulmonary aspergillosis are available. The basic procedure is the fungal culture of respiratory secretions. Repeated positive culture from the lower respiratory tract is supportive of IPA. [10] Multiple signs have been described radiologically as being associated with IPA, such as consolidation, nodules, the “halo sign,” and the “air-crescent sign,” and the last two signs are pathognomonic of IPA. The halo sign corresponds to a low attenuation surrounding a mass or nodule, and the air-crescent sign is related to the contraction of infarcted lung tissue delineated by an air-filled space. [11] Serological methods such as the detection of the antigen galactomannan (cell wall glycoprotein) by ELISA, EIA, or immunoblot test (sensitivity and specificity >90%) are used. However, it may be positive even in patients with treated with piperacillin/tazobactum and cross reaction may be seen with fungi such as Cryptococcus species, Histoplasma species, and cyclophosphamide. [12] Hence, antigenemia cannot be considered diagnostic criteria alone and the polymerase chain reaction for the detection of DNA and RNA copies are being increasingly used from bronchoalveolar lavage. [13] [Table 1] summarizes the various diagnostic tests [14] available for IPA. However, the cytological/histopathological demonstration of typical septate hyphae or growth of Aspergillus in tissue specimens remains the gold standard for a definitive diagnosis as reported in the above cases. In conclusion, the present case gives an importance to the need of fungal culture for the lower respiratory tract sample in the case of acute pulmonary symptoms in immunocompetent individuals with no underlying pulmonary disease. If the patients are farmworkers, then the probability of aspergillosis is very likely. Thus, these cases can be considered as eye openers for considering pulmonary aspergillosis in immunocompetent farmworkers with acute pulmonary symptoms as well.
| Table 1: Definitions of invasive pulmonary aspergillosis
Click here to view |
| References |
| 1. | Patterson TF. “Aspergillus species”. In: Mandell GL, Bennett JE, Dolin R, editors. Mandell, Douglas, and Bennett’s principles and practice of infectious diseases. 7 th ed., vol. 2., Chap. 258. Philadelphia, PA (U.S.A.): Churchill Livingstone Elsevier; 2010. p. 3241-55. |
| 2. | Patterson TF, Kirkpatrick WR, White M, Hiemenz JW, Wingard JR, Dupont B, et al. Invasive aspergillosis. Disease spectrum, treatment practices, and outcomes. I3 Aspergillus Study Group. Medicine (Baltimore) 2000;79:250-60. |
| 3. | Meyer RD. Aspergillus species. In: Gorbach SL, Bartlett JG, Blacklow NR, editors. Infectious Diseases. 3 rd ed., chap. 269. Philadephia: W. B. Saunders Lippincott Williams & Wilkins; 2004. p. 2212-8. |
| 4. | Kang EY, Kim DH, Woo OH, Choi JA, Oh YW, Kim CH. Pulmonary aspergillosis in immunocompetent hosts without underlying lesion of the lung: Radiologic and pathologic findings. AJR Am J Roentgenol 2002;178;1395-9. |
| 5. | Ellis RH. Pulmonary aspergillosis in two families. Postgrad Med J 1968;44:178-94. |
| 6. | Ruhnke M, Eichenauer E, Searle J, Lippek F. Fulminant tracheobronchial and pulmonary aspergillosis complicating imported Plasmodium falciparum malaria in an apparently immunocompetent woman. Clin Infect Dis 2000;30:938-40. |
| 7. | Oh HJ, Kim HR, Hwang KE, Kim YS, Ahn SH, Yang SH, et al. Case of pseudomembranous necrotizing tracheobronchial aspergillosis in an immunocompetent host. Korean J Intern Med 2006;21:279-82. |
| 8. | Unis GU, Picon PD, Severo LC. Coexistence of intracavitatory fungal colonization (Fungal ball) and active tuberculosis. J Bras Pneumol 2005;31:139-43. |
| 9. | Baradkar VP, Mathur M, Kumar S. Uncommon presentation of pulmonary aspergilloma. Indian J Med Microbiol 2009;27:270-2. |
| 10. | Greub G, Bille J. Aspergillus species isolated from clinical specimens: Suggested clinical and microbiological criteria to determine significance. Clin Microbiol Infect 1998;4:710-6. |
| 11. | Kuhlman JE, Fishman EK, Burch PA, Karp JE, Zerhouni EA, Siegelman SS. CT of invasive pulmonary aspergillosis. AJR Am J Roentgenol 1988;150:1015-20. |
| 12. | Viscoli C, Machetti M, Cappellano P, Bucci B, Bruzzi P, Van Lint MT, et al. False-positive galactomannan platelia Aspergillus test results for patients receiving piperacillin-tazobactum. Clin Infect Dis 2004;38:913-6. |
| 13. | Musher B, Fredricks D, Leisenring W, Balajee SA, Smith C, Marr KA. Aspergillus galactomannan enzyme immunoassay and quantitative PCR for diagnosis of invasive Aspergillosis with Bronchoalveolar lavage fluid. J Clin Microbiol 2004;42:5517-22. |
| 14. | Bulpa P, Dive A, SibilleY. Invasive pulmonary aspergillosis in patients with chronic obstructive pulmonary disease. Eur Respir J 2007;30:782-800. |
Source of Support: None, Conflict of Interest: None
| Check |
DOI: 10.4103/1755-6783.120998
| Figures |
[Figure 1], [Figure 2], [Figure 3]
| Tables |
[Table 1]



