Aims: This study was aimed to study the clinical and radiological profile of neurocysticercosis and outcomes of treatment in neurocysticercosis. Settings and Design: A prospective observational study in a tertiary care hospital. Methods and Material: A total of 40 patients coming under definitive or probable diagnosis of neurocysticercosis as per the revised diagnostic criteria for neurocysticercosis were included in this study. Clinical history, physical examination, and neuroimaging (CT or MRI) studies were done at the beginning. Patients were treated with albendazole, corticosteroids, and antiepileptic drugs. They were followed up clinically for 3 months and neuroimaging study was repeated 3 months after albendazole therapy. Results: Definitive cases were 28 (70%) and probable cases were 12 (30%). Male: female ratio was 2.07. Mean age of 29.62 ± 9.08 years. The commonest presentation was seizures in 38 (95%). Twenty-nine (72.5%) patients had a single lesion and 11 (27.5%) had multiple lesions in initial imaging study. Commonest site of lesion was parietal lobe (45%). Most common stage of presentation was colloidal (55%). Thirty-seven (92.5%) patients were free of seizures at the end of three months. At the end of 3 months, 23 (57.5%) patients had radiological resolution (Complete resolution + partial resolution + calcification). Conclusions: Neurocysticercosis usually affects young persons, mostly in third decade, being equally common among vegetarians. Most common clinical manifestation is seizures. Single lesion is more common than multiple lesions, commonly presenting in colloidal. Clinical and radiological response to 4-week therapy with albendazole is quite satisfactory. All cases of young- and adult-onset epilepsy in tropical countries should be investigated for neurocysticercosis. Keywords: Albendazole, colloidal stage, neurocysticercosis, seizures
Neurocysticercosis is the commonest parasitic disease of nervous system. [1] The age-adjusted prevalence of active epilepsy in tropical countries ranges from 10 to 15 per 1 000 inhabitants, almost twice the level in western countries. [2] Neurocysticercosis has been estimated to cause at least 50 000 deaths worldwide annually. [2] In India, neurocysticercosis has been identified as a cause of 2.2 to 6% unselected cases of seizures. [3] Neurocysticercosis is caused by the larval stage (cysticerci) of the pork tapeworm Taenia solium. Clinical manifestations of neurocysticercosis are varied due to individual differences in the number, size, and topography of lesions and in the severity of the host’s immune response to the parasites. This study aimed to study the clinical and radiological profile of neurocysticercosis and outcomes of treatment in neurocysticercosis.
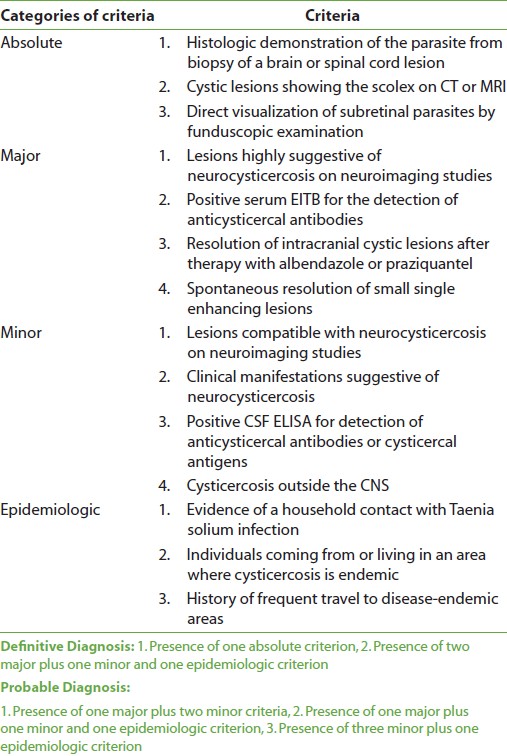
This was a prospective observational study conducted over a period of one year from June 2008 to June 2009, and included 40 patients. Inclusion criteria Patients coming under definitive or probable diagnosis of neurocysticercosis as per the revised diagnostic criteria for neurocysticercosis [4] [Table 1] were included in this study. However, histologic demonstration of the parasite from biopsy of a brain lesion was not done, as this is an invasive procedure. Also, serum EITB and CSF ELISA tests were not done due to nonavailability of these tests in the institute and non-affordability of the patients. Thus, the diagnosis was based on clinical and radiological features.
Exclusion criteria Patients of tuberculosis, HIV-reactive patients, and patients with a known malignancy were excluded due to a greater chance of intracranial space-occupying lesions in these patients. A detailed medical history with emphasis on the description of the seizure, general examination, and neurological examination was performed. Diagnostic evaluation included hemoglobin, peripheral blood smear, total and differential leukocyte counts, microscopic examination of stool done thrice for taeniasis, work-up for tuberculosis (erythrocyte sedimentation rate, Mantoux test, chest radiograph), HIV I and II, contrast-enhanced CT scan of brain, or MRI brain. Number, site, stage, size of the lesions, presence of scolex, and perilesional edema were noted. All patients received albendazole in a dose of 15 mg/kg body weight/day in three divided doses for 28 days and oral prednisolone 2 mg/kg/day for initial 5 days of therapy. Antiepileptic drugs were given. Patients were followed up regularly for 3 months after completion of Albendazole therapy. Imaging study was repeated after three months after initiation of Albendazole therapy. Following were considered indicators of therapeutic outcome: Radiological outcome:
Clinical outcome was measured in terms of recurrence of seizures, based on objective reports from patients.
There were 40 patients included in the study, of which definitive cases were 28 (70%) and probable cases were 12 (30%). Distribution of cases as per criteria for diagnosis of neurocysticercosis is shown in [Table 2].
Patient characteristics Total 40 patients were included in this study, of which 27 (67.5%) patients were males and 13 (32.5%) were females. The male: female ratio was 2.07. The subject age varied from 16 to 55 years with a mean age of 29.62 ± 9.08 years. Maximum patients were in the age group of 21 to 30 years (45%), followed by 31 to 40 years (25%), 11 to 20 years (15%), 41 to 50 years (12.5%), and >50 years (2.5%). Majority (50%) of cases were of lower socioeconomic strata, followed by upper lower (27.5%), lower middle (15%), upper middle (5%), and upper class (2.5%). Nineteen patients (47.5%) were pure vegetarians and 21 (52.5%) patients took a mixed diet. None of the patients were pork-eaters. None of the patients had domestic or occupational exposure to pigs. Of 40, 26 (65%) patients were rural and 14 (35%) patients were urban. The presenting clinical features are shown in [Table 3]. There were 19 (47.5%) patients who had simple partial seizures, 3 (7.5%) patients had complex partial seizures, 4 (10%) had partial seizures with secondary generalization, and 14 (35%) patients had primarily generalized seizures. In 6 (15%) patients, Jacksonian progression of seizure activity was seen. Only 5 (12.5%) patients had postictal weakness (Todd’s paresis) lasting for few minutes to 8 hours. Status epilepticus was present in 3 (7.5%) patients. Papilledema was observed in 4 (10%) patients. Ocular cysticercosis was not seen in any patient.
Investigations Mean hemoglobin level was 10.66 ± 2.54 g/dl. Eosinophilic percentage was raised (>5%) in 4 (10%) patients. Stool microscopy for taeniasis was negative in all patients. Characteristics of first CT scan/MRI Thirty-five (87.5%) patients underwent contrast-enhanced CT scan and 5 (12.5%) patients underwent MRI scan at the beginning of the study. Of 40 patients, 29 (72.5%) patients had a single lesion and 11 (27.5%) had multiple lesions in initial imaging study. Commonest site of lesion was parietal lobe (45%). Eleven patients (27.5%) had lesions involving multiple lobes and 9 (22.5%) patients had frontal lobe lesions. Only one patient each had a lesion in temporal and occipital lobes. Most common stage of presentation was colloidal (55%), followed by vesicular (12.5%) and granular-nodular (5%), whereas 27.5% patients had lesions in multiple stages of development. Of 40 patients, 24 (60%) patients were found to have lesions ≤10 mm, whereas 16 (40%) patients had lesions >10 mm. Scolex was seen in initial imaging study in 15 (37.5%) patients. Twenty-six (65%) patients had perilesional edema at the time of presentation. Perilesional edema was ≤5 mm in 24 (60%) patients and >5 mm in 2 (5%) patients, whereas 14 (35%) patients had no perilesional edema. None of the patient had mass effect due to lesion. Extraneural cysticercosis Two (5%) patients had evidence of extraneural cysticercosis. One patient had palpable muscle nodule on anterior abdominal wall which was proved to be cysticercosis on aspiration cytology. Other patient was found to have vesicular cysticercal lesions with central scolex in tongue muscles and paravertebral muscles of neck on MRI scan. Clinical outcome Clinical outcome was assessed on the basis of recurrence of seizures, based on objective reports from patients. Thirty-seven (92.5%) patients were free of seizures, whereas three patients (7.5%) had recurrence of seizures, at the end of three months. All 29 patients who had a single lesion in the initial imaging study were seizure free and eight of 11 patients with multiple lesions in initial imaging study were free of seizures at the end of 3 months. Of the 29 patients with a single lesion who were seizure free, 13 (44.82%) had complete resolution of the lesion on follow-up imaging study at the end of 3 months; 04 (13.79%) had partial resolution; 02 (6.89%) lesions underwent calcification; and lesions remained unaltered in 10 (34.48%) patients. However, of eight patients with multiple lesions who were seizure free, 3 (37.5%) had partial resolution and 1(12.5%) patient had calcification of lesion. Three (7.5%) patients had recurrence of seizures during the three-month follow-up period. All of these patients had multiple lesions in the initial imaging study and the lesions were unaltered in the follow-up study at the end of 3 months. No patient developed signs of elevated intracranial pressure during or soon after therapy. Six patients (15%) developed headache on day 3 to 4 of treatment with cysticidal drugs not associated with any other feature of raised intracranial pressure. Interruption of the therapy was not required in any of these cases. Radiological outcome Radiological follow-up at 3 months in patients with single lesion (n = 29) and in patients with multiple lesions (n = 11) is shown in [Table 4] and [Table 5], respectively. At the end of 3 months, only one patient (2.5%) had perilesional edema >5 mm and 3 (7.5%) patients had perilesional edema ≤5 mm. None of the patients had shown appearance of new lesions on follow-up imaging study.
The subject age varied from 16 to 55 years with a mean age of 29.62 ± 9.08 years. Maximum patients were in the age group of 21 to 30 years. Mean age of patients in other studies across the globe was 33 years (Garcia et al); [5] 27 ± 18 years (Carpio and Hauser). [6] In adult case series from India, the mean age of patients were 29.1 years (Das et al.); [3] 28 ± 12 years with range of 12 to 66 years (Kaushal et al.). [7] These data show that neurocysticercosis is common in children and young adults worldwide. Thus, the age distribution of the patients in the present study was similar to other Indian as well global studies. Cysticercosis is generally a disease of lower socioeconomic conditions associated with poor hygiene and sanitation. In a study from Kerala, Kuruvilla et al.[8] found that 73% patients belonged to lower socioeconomic category. In other studies from Chandigarh, 69% and 82.7% of the patients of lower socioeconomic strata were involved. [9],[10] Majority (77.5%) of the cases in the present study were of lower socioeconomic class. Thus, results were similar to other Indian studies. Although cysticercosis had been reported to be higher in nonvegetarian people, especially pork eaters, many (46%) of our patients were pure vegetarians, and none of the patients consumed pork, thereby supporting the fact that feco-oral contamination seems to be the major route of the disease. In a previous study from Chandigarh, 53.2% patients were documented as pure vegetarians. [10] In the study from China, Chaoshuang et al.[11] found that urban population is at more risk than rural population. The percentages of urban patients in two Brazilian studies were 86.2% (Chagas et al.) [12] and 93.8% (Benedeti et al.). [13] Rajshekhar et al., [14] who studied active epilepsy as an index of burden of neurocysticercosis in Vellore district, India, found that the prevalence in urban clusters was more than twice in the rural cluster (6.23 vs 3.04 per 1 000). Contrary to the current literature, the present study has a high percentage of rural patients, probably related to poor hygienic conditions and higher amount of fecal contamination of drinking water in rural areas. Seizures are reported as the most common symptom in neurocysticercosis, occurring in 70 to 90% of patients. Similarly, 38 (95%) patients in the current study presented with seizure as the chief complaint. In literature, partial seizures are the most commonly reported seizures in patients with single lesion neurocysticercosis. [10],[15],[16],[17] Majority (86.20%) of the patients with single lesion in this study also had partial seizures. Papilledema was observed in 4 (8%) patients. Das et al. reported papilledema in 16.5% patients. [3] Most of the studies in India deal with single lesion neurocysticercosis (solitary cysticercal granuloma). A study by Kuruvilla et al.[8] found single lesion in 40% and multiple lesions in 60% patients. Single lesion was present in 76% and multiple lesions in 24% patients in a study by Singhi et al. [16] Similarly, Kotokey et al.[18] reported single lesion in 66.66% and multiple lesions in 33.33% patients; and Rajshekhar et al.[14] reported single lesion in 60.88% and multiple lesions in 39.13% patients. Thus, although most of the Latin American studies show that multiple lesion neurocysticercosis is common in that part of the world, most of Indian studies show that single lesion neurocysticercosis is more common form of disease in India. The present study found single lesion in 72.5% patients and multiple lesions in 27.5% patients, showing that single lesion neurocysticercosis is more common than multiple lesions, which is in agreement with most other Indian studies. Parietal lobe has been found to be the most common site of single lesion neurocysticercosis. [9],[10],[15],[19] In this study, 45% of the lesions were situated in the parietal lobe. Baranwal et al. and Singhi et al. reported parietal lobe involvement in 41% and 57.3% patients, respectively. [9],[10] Since most of the patients present with motor or sensorimotor seizures, distribution of lesions about the sensorimotor cortex is well expected. Less commonly reported sites are temporal lobe, basal ganglion, and cerebellum, which was also observed in our study. [9],[10] Thus, the distribution of the lesion in this study was similar to other studies. The most common stage of presentation was colloidal (55%). This can be explained by the fact that in colloidal stage, there is degeneration of cyst wall and active inflammation leading to seizures. In vesicular phase, there is little or no evidence of perilesional inflammation and the cellular immune response is suppressed leaving these viable cysts asymptomatic for long. Thus, only 12.5% patients presented in vesicular phase. Neurocysticercosis lesions usually measure 20 mm or less in its maximal dimension. Majority (60%) of the patients were found to have maximum lesion diameter ≤10 mm. The visualization of a scolex on CT scan confirms the diagnosis of neurocysticercosis. However, it is not seen in all the cases. It could be visualized in only 37.5% patients. Similar figures have been reported previously also. [19] Thirty-seven (92.5%) patients were free of seizures, whereas three patients (7.5%) had recurrence of seizures, at the end of three months. The seizure recurrence in patients treated with Albendazole in other studies was 13% (Gogia et al.), [15] 56% (Garcia et al.), [5] 13% (Kalra et al.), [20] and 18% (Kuruvilla et al.). [8] Thus, the seizure recurrence of 7.5% in the current study is less than that observed by other authors. In this study, of 40 patients, 23 (57.5%) patients had radiological resolution (complete resolution + partial resolution + calcification) at the end of three months. The radiological resolution in various other studies at the end of three months was 88% (Padma et al. ), [21] 87% (Padma et al.), [22] 64.5% (Baranwal et al.), [9] 62.5% (Singhi et al.), [23] and 60% (Sotelo et al). [24] The radiological resolution in 57.5% in the current study is comparable with last three studies.
This study concludes that neurocysticercosis usually affects young persons, mostly in third decade. It is equally common among vegetarians. Most common clinical manifestation is seizures, partial seizures being most common in patients with single lesion and primarily generalized seizures in patients with multiple lesions. Single lesion is more common than multiple lesions, commonly presenting in colloidal stage (ring-enhancing lesion). Clinical and radiological response to 4-week therapy with albendazole is quite satisfactory, with control of seizures in 92.5% patients and radiological resolution in 57.5% patients at the end of 3 months. We recommend that all cases of young- and adult-onset epilepsy in tropical countries should be investigated for neurocysticercosis.
Source of Support: None, Conflict of Interest: None
[Table 1], [Table 2], [Table 3], [Table 4], [Table 5] |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||